Overview
Scientific writing and publishing form the heart of medical research. A well-written paper not only shares data but also tells a story of discovery.
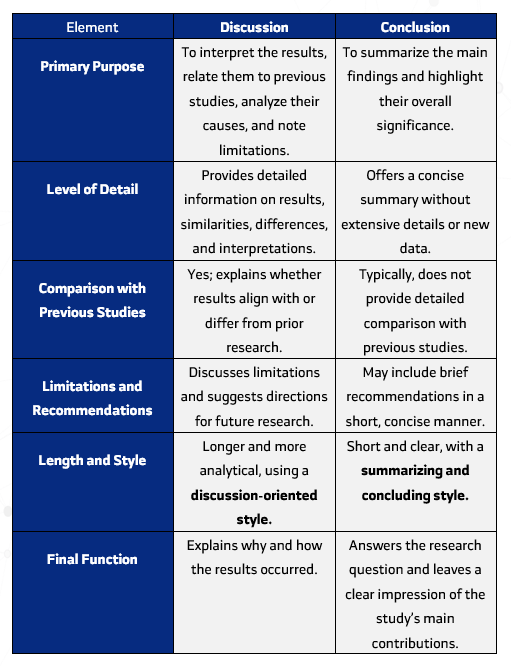
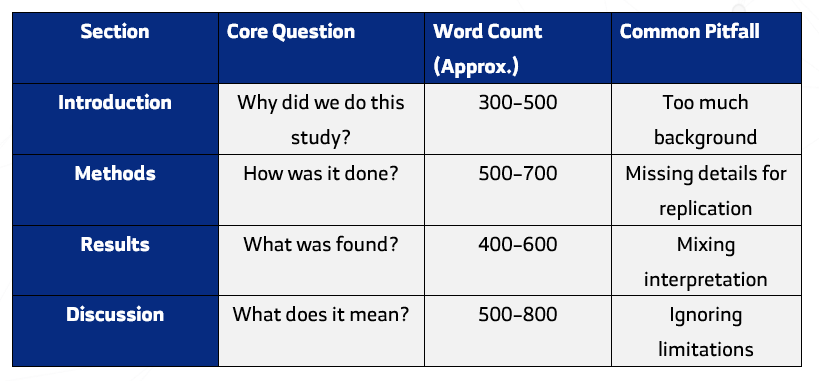
Most journals follow a standard format known as IMRaD — Introduction, Methods, Results, and Discussion.
Scientific Writing Using the IMRaD Model is a structured approach that forms the foundation of modern medical research, and is widely used as a standard framework for presenting studies in a clear and organized manner. Understanding this structure helps young researchers organize ideas clearly and meet publication standards
The IMRaD Format: Your Paper’s Backbone
Scientific writing using the IMRaD model guides the reader step by step through the research: What is the topic? How was the study conducted? What are the findings? And what do these findings mean within the context of current scientific knowledge?
Each of these sections will be discussed in detail.
| Section | Question Answered | Purpose |
|---|---|---|
| Introduction | Why was the study done? | Provides context and sets the objective. |
| Methods | How was it done? | Describes how the study was conducted. |
| Results | What was found? | Presents findings objectively. |
| Discussion | What do the results mean? | Interprets and relates them to previous work. |
Writing a Compelling Title and Abstract
Your title and abstract are the first impression editors and readers get, and often decide whether your paper is read or ignored. Therefore, crafting these elements with precision and professionalism is a critical step in the scientific publishing process.
The Title
The title is the first element visible in search engines and academic journals. It should be direct, concise, informative, and search-friendly. Its purpose is to convey the main idea of the research without ambiguity or exaggeration, while ensuring discoverability in electronic searches.
The Abstract
General Tips for Writing an Effective Title:
- Use clear keywords that accurately reflect the study topic.
- Make the title descriptive rather than general; avoid vague titles such as “A Study on Cancer”.
- Ideally, the title should consist of 8–15 words, though some journals allow up to 20 words, especially in clinical articles—for example: “The Association Between Smoking Habits and Lung Cancer Incidence Among Young Adults in Urban Areas.”
- Do not include study results in the title.
- Include the main variable, population, and the study design when relevant, such as Cross-sectional Study or Randomized Controlled Trial.
Example:
❌
“A study of diabetes”
✅
“Glycemic control among type 2 diabetes patients attending primary care clinics in Amman: a cross-sectional study.”
The abstract is the most frequently read section of a research paper. Its purpose is to provide a concise and comprehensive overview of the study’s objectives, methodology, results, and key conclusions, allowing readers to assess the relevance and significance of the research before reading the full paper
Typical Structure of a Scientific Abstract:
- Background: A brief overview of the problem or phenomenon under investigation.
- Objective: The aim or research question the study seeks to address.
- Methods: The study type, sample, instruments, and data analysis approach.
- Results: A concise presentation of the main statistical or numerical findings.
- Conclusion: Interpretation of the results and their significance.
Example:
Lung cancer is among the most prevalent types of cancer worldwide, with smoking being the primary risk factor. This study aimed to analyze the relationship between smoking and lung cancer risk in adults. A cross-sectional study was conducted with 500 participants, collecting data via questionnaires and medical record analysis. Results indicated that smokers had a threefold higher risk of developing lung cancer compared to non-smokers. These findings highlight the importance of strengthening smoking cessation awareness programs as an effective preventive measure against lung cancer.
General Tips for Writing an Abstract:
- Write the abstract after completing the full study to ensure accuracy.
- Use formal and clear language without personal opinions or emotional expressions.
- Avoid including references, tables, or figures in the abstract.
- Adhere to the word limit specified by the publisher (typically 150–250 words).
- Ensure that every piece of information in the abstract is present in the main text of the paper.
The Introduction
The introduction of any scientific paper aims to convince the reader that the study is necessary and addresses a genuine knowledge gap. It prepares the reader to understand why the research was conducted, not just what was investigated. The introduction should be brief (3–5 paragraphs), and is typically presented in a sequential and logical manner, covering:
The introduction can be likened to a funnel, starting from a broad concept and gradually narrowing down to the specific research objective.
Within this framework, scientific writing using the IMRaD model provides a logical structure that helps researchers present the background, define the knowledge gap, and clearly state the objective and hypothesis.
Background
Knowledge Gap
This paragraph highlights what previous studies have not addressed or what remains unknown. It can reference prior research and then identify areas of deficiency or uncertainty.
The introduction typically concludes with a clear statement of the study’s purpose or main hypothesis, preferably in a direct and precise sentence.
Objective and Hypothesis
The objective and hypothesis are essential elements of any scientific study, guiding the research from inception to conclusion. Without a clear objective and defined hypothesis, a study loses direction and cannot have its results evaluated scientifically.
The Hypothesis
The hypothesis is a scientific prediction based on existing knowledge about what the study’s results might reveal. It is a logical assumption grounded in available evidence and theories, which the researcher aims to test through the study. A valid hypothesis should be:
- Testable: Can be evaluated through data collection and analysis.
- Scientifically grounded: Based on evidence and reasoning, not mere opinion.
- Falsifiable: Can be proven true or false through the study’s results.
Relationship Between Objective and Hypothesis
The objective and hypothesis are logically connected:
- The objective defines what is being investigated.
- The hypothesis predicts what is expected to be discovered.
The objective provides the overall framework, while the hypothesis offers a specific, testable application within that framework. Once clearly formulated, they facilitate the development of the Methods section and the selection of appropriate tools and analyses.
The Methods
The Methods section describes how the study was conducted so that any other researcher can reproduce it and verify the results “Reproducibility”. The researcher should provide a clear and precise description of all steps and procedures, avoiding vague or incomplete details. This section is among the most technical and organized parts of a scientific paper and should be written in past tense, as the events have already been completed.
This level of clarity and transparency is essential in scientific writing using the IMRaD model, allowing other researchers to evaluate and reproduce the study reliably.
Main Components:
This section is usually organized into subsections including:
- Study Design
- Setting & Duration
- Participants & Sample Size
- Inclusion & Exclusion Criteria
- Procedures & Data Collection
- Statistical Analysis
- Ethical Considerations
Study Design
The researcher must clearly specify the type of study, such as:
- Descriptive Study
- Case–Control Study
- Prospective Cohort Study
- Randomized Clinical Trial
For further details, please refer to our article:
From idea to design: A simple Guide to choosing your Research Methodology
Setting & Duration
This subsection provides information about where the study was conducted and over what period.
Participants & Sample Size
▹ Participants: Describe the individuals or cases included in the study, including:
- Total number of participants
- Age, gender, and geographic location
- Selection method (random, voluntary, hospital-based, school-based, etc.)
- Any special characteristics relevant to the research (e.g., patients, students, smokers)
▹ Sample Size: The actual number of participants or units included. Sample size should be determined using statistical formulas or software to ensure adequate power to detect real differences. Report:
- Final sample size
- Statistical rationale for selection
▹ There are many tools that can help you with the sample size like ResRef Sample Size Calculator on our website.
Inclusion & Exclusion Criteria
These criteria define who can participate and who should be excluded, ensuring accuracy and reducing bias.
- Inclusion Criteria: Required characteristics for eligibility, e.g., age, gender, health status, location.
- Exclusion Criteria: Factors that prevent participation because they may affect results or confound data.
The goal is to select a homogeneous and representative sample, ensuring reliable and reproducible results.
Procedures & Data Collection
Statistical Analysis
This subsection explains how data were analyzed to derive results and understand relationships between variables. It typically includes:
- Software used (e.g., SPSS, R, Excel)
- Statistical tests applied (e.g., Chi-square test, Logistic Regression)
- Significance level (p-value) to determine scientific relevance
- Methods for presenting results (mean, standard deviation, percentages, confidence intervals)
Ethical Considerations
Adherence to ethical principles is essential, especially in studies involving human participants or personal data. This section ensures the study respected participants’ rights and followed internationally recognized ethical standards.
The researcher should specify:
- Approval from an ethics committee (Ethical Approval)
- Written informed consent from all participants (Informed Consent) after explaining study objectives and participation details
- Confidentiality of all data without revealing personal identities
- Voluntary participation, allowing withdrawal at any time without consequences
The Results
The results section presents the study’s findings clearly and objectively, without interpretation or discussion (interpretation is reserved for the discussion section). Results should be organized so that readers can easily understand. Use tables and figures to make data easy to grasp.
Results section includes:
- Participant and group distribution
- Primary and secondary outcomes
- Statistical analyses applied
- Data presentation through text, tables, and figure
The Discussion
The Discussion is where the researcher interprets the results within the broader scientific context. It goes beyond presenting findings by connecting them to existing theories and previous studies.
It can be structured as follows:
- Summary of key findings
- Begin by highlighting the main results without repeating all numbers.
- Focus on points that support the research hypothesis or objectives.
- Comparison with previous studies
- Explain whether the results are consistent with prior research.
- If differences exist, discuss potential reasons (e.g., sample size, measurement methods, study conditions).
- Interpretation of results
- Offer explanations for the findings, linking them to possible factors or mechanisms.
- Connect interpretations to theories or scientific background described in the Introduction.
- Limitations
- Describe any constraints that may affect the results, such as small sample size, short study duration, or measurement errors.
- Future directions
- Provide recommendations for future research that may address current limitations or expand knowledge on the topic.
The Conclusion
The conclusion summarizes the main findings and highlights their significance. It should:
- Avoid presenting new data
- Address the primary research question
- Provide readers with a clear understanding of what the study achieved and its importance
- Exclude detailed repetition of results
Keep it short, 2–3 lines highlighting the main takeaway.
Common Mistakes
- Writing results in the Introduction or vice versa.
- Using jargon or overly complex sentences.
- Ignoring journal author guidelines.
- Forgetting ethical approval or participant consent.
- Overstating conclusions beyond the data.
Key Takeaways
- The IMRaD format helps you present research logically and clearly.
- Focus on clarity, accuracy, and transparency in every section.
- Use the title and abstract to capture attention.
- Keep methods reproducible and results factual.
- In discussion, interpret — don’t repeat — your results.
- Always proofread and follow journal guidelines before submission.
A Word From ResRef
At ResRef, we believe that mastering scientific writing is essential for transforming research into meaningful contributions to global knowledge. Scientific writing using the IMRaD model provides researchers with a clear, structured, and universally accepted framework that enhances clarity, reproducibility, and academic credibility. Whether you are a medical student, early-career researcher, or experienced academic, understanding and applying this structure will strengthen your ability to communicate findings effectively and improve your chances of successful publication. Our mission is to equip researchers with practical, accessible guidance that empowers them to write with confidence and contribute to evidence-based science.
Frequently Asked Questions (FAQs)
1. How does scientific writing using the IMRaD model improve research quality?
Scientific writing using the IMRaD model improves research quality by enforcing clarity, structure, and methodological transparency. It ensures that each part of the research process is documented systematically, allowing readers and reviewers to assess the validity of the study. This structured approach also reduces ambiguity, strengthens scientific credibility, and increases the likelihood of acceptance in peer-reviewed journals.
2. Do all scientific journals require the IMRaD format?
Most medical, clinical, and scientific journals use the IMRaD format or a closely related structure. While some variations exist depending on the journal and study type, the fundamental principles of Introduction, Methods, Results, and Discussion remain widely accepted across disciplines.
3. What are the most common mistakes researchers make when using the IMRaD structure?
Common mistakes include mixing results with interpretation, providing insufficient detail in the Methods section, writing overly long or unfocused introductions, and drawing conclusions that are not fully supported by the data. Another frequent error is failing to clearly define the research objective or knowledge gap. Avoiding these mistakes helps ensure clarity, reproducibility, and stronger scientific impact.
References
- Grimes, D. A., & Schulz, K. F. (2002). Descriptive studies: what they can and cannot do. The Lancet, 359(9301), 145–149.
- Vandenbroucke, J. P., & Pearce, N. (2012). Case–control studies: basic concepts. International Journal of Epidemiology, 41(5), 1480–1489.
- Hulley, S. B., Cummings, S. R., Browner, W. S., Grady, D. G., & Newman, T. B. (2013). Designing Clinical Research (4th ed.). Lippincott Williams & Wilkins.
- National Cancer Institute. (2022). Lung Cancer Prevention (PDQ®)–Health Professional Version.
- Doll, R., & Hill, A. B. (1956). Lung cancer and other causes of death in relation to smoking. British Medical Journal, 2(5001), 1071–1081.
- Thun, M. J., Carter, B. D., Feskanich, D., Freedman, N. D., Prentice, R., Lopez, A. D., … & Gapstur, S. M. (2013). 50-year trends in smoking-related mortality in the United States. New England Journal of Medicine, 368(4), 351–364.
- Hartley, J. (2019). Current findings from research on structured abstracts. Journal of the Medical Library Association, 107(1), 97–100.
- Pautasso, M. (2013). Ten simple rules for writing a literature review. PLoS Computational Biology, 9(7), e1003149.
- Creswell, J. W., & Creswell, J. D. (2018). Research design: Qualitative, quantitative, and mixed methods approaches (5th ed.). Sage Publications.
- Day, R. A., & Gastel, B. (2022). How to Write and Publish a Scientific Paper (9th ed.). Cambridge University Press.
Authorship and Contributions
The following section acknowledges the individuals who contributed to the authorship, editing, translation, and preparation of this article, ensuring its academic integrity and clarity.

Dr. Lama Aladal
Author

Prof. Hamoud Hamed
Editor
Professor of General Surgery and Laparoscopic Surgery, Faculty of Medicine, Damascus University.

Prof. Hamoud Hamed
Editor

Dr. Haidar Alsakher
Translator & Formatter
A sixth-year medical student at the Faculty of Medicine, Damascus University, contributes to the Educational and Web departments at ResRef.

Dr. Haidar Alsakher
Translator & Formatter